-
Home
-
About JCTR
-
Gold Open Access
-
Issues
-
Editorial board
-
Author guidelines
-
Publication fees
-
Online first
-
Special issues
-
News
-
Publication ethics
-
Partners
-
Submit your manuscript
-
Submit your review report
-
Editorial Office
-

This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License. ISSN print: 2382-6533 ISSN online: 2424-810X
Volume 8 Issue 6
Bone marrow mesenchymal stem cell-derived exosomal LINC00847 inhibits the proliferation, migration, and invasion of Ewing sarcoma
Lu Huang†, Jiachao Xiong†, Jimin Fu†, Zhenhai Zhou, Honggui Yu, Jiang Xu, Liang Wu, Kai Cao*
Huang et al. J Clin Transl Res 2022; 8(6):15
Published online: November 24, 2022
Abstract
Background: Ewing sarcoma (ES) is one of the most lethal primary bone tumors with a poor survival rate. Current evidence suggests that extracellular vesicles (EVs) derived from bone marrow mesenchymal stem cells (BMSCs) loaded with abundant biological functional lncRNAs confer therapeutic benefits against the development of various tumors.
Aim: This study aimed to investigate the role of exosomal lncRNAs from BMSCs in the pathogenesis of ES.
Methods: Bioinformatic analysis and qRT-PCR experiments were used to detect the expression level of LINC00847 in ES tissues and cells. Cell biology experiments examined the effect of in vitro proliferation, migration, and invasion abilities and the biological function of BMSCs-derived LINC00847. Finally, we constructed a LINC00847-associated competitive endogenous RNA (ceRNA) network by in silico methods. Gene Set Enrichment Analysis (GSEA) was conducted to reveal the potential molecular mechanism of LINC00847.
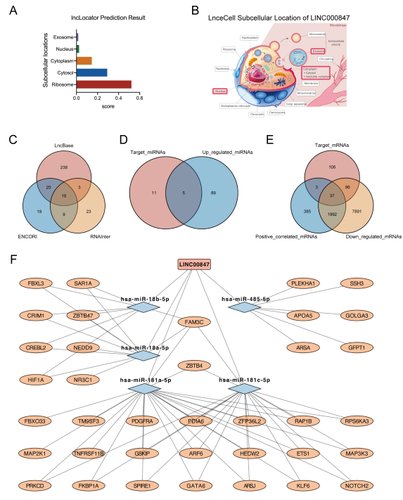
Results: We found that LINC00847 was markedly downregulated in ES. Overexpression of LINC00847 inhibited ES cell proliferation, migration, and invasion. Furthermore, BMSCs-derived EVs inhibited the proliferation, migration, and invasion of ES cells by delivering LINC00847. We constructed a LINC00847 related-ceRNA network contains five miRNAs (miR-18a-5p, miR-18b-5p, miR-181a-5p, miR-181c-5p, and miR-485-3p) and four mRNAs (GFPT1, HIF1A, NEDD9, and NOTCH2).
Conclusions: Overall, this study found that BMSCs-EVs-derived exosomal LINC00847 inhibited ES cell proliferation, migration, and invasion. The ceRNA regulatory mechanism of LINC00847 may participate in the pathogenesis of the malignant phenotype of Ewing sarcoma.
Relevance for patients: These findings suggest that BMSCs-derived exosomal lncRNAs may be used for the personalized treatment of tumors, providing a novel theoretical framework for treating ES.
†These authors contributed equally
DOI: http://dx.doi.org/10.18053/jctres.08.202206.015
Author affiliation
1. The Maternal and Children Health Hospital of Jiangxi Province, 318 Bayi Avenue, Nanchang, Jiangxi Province, 330006, China
2. The Orthopedics Hospital, The First Affiliated Hospital of Nanchang University, 1519 Dongyue Avenue, Nanchang County, Nanchang, Jiangxi Province, 330200, Chin
*Corresponding author
Kai Cao
The Orthopedics Hospital, The First Affiliated Hospital of Nanchang University, Nanchang, Jiangxi Province, 330200, PR China.
E-mail: kaichaw@126.com
Handling editor:
Michal Heger
Department of Pharmaceutics, Utrecht University, the Netherlands
Department of Pharmaceutics, Jiaxing University Medical College, Zhejiang, China

