-
Home
-
About JCTR
-
Gold Open Access
-
Issues
-
Editorial board
-
Author guidelines
-
Publication fees
-
Online first
-
Special issues
-
News
-
Publication ethics
-
Partners
-
Submit your manuscript
-
Submit your review report
-
Editorial Office
-

This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License. ISSN print: 2382-6533 ISSN online: 2424-810X
Volume 4 Issue 1
Post-hepatectomy liver regeneration in the context of bile acid homeostasis and the gut-liver signaling axis
Lianne de Haan, Sarah van der Lely, Anne-Loes K. Warps, Quincy Hofsink, Pim B. Olthof, Mark J. de Keijzer, Daniël A. Lionarons, Lionel Mendes-Dias, Bote G. Bruinsma, Korkut Uygun, Hartmut Jaeschke, Geoffrey C. Farrell, Narci Teoh, Rowan F. van Golen, Tiangang Li, Michal Heger
de Haan et al., J Clin Transl Res, 2018, 4(1): 1
Published online: February 16, 2018
Abstract
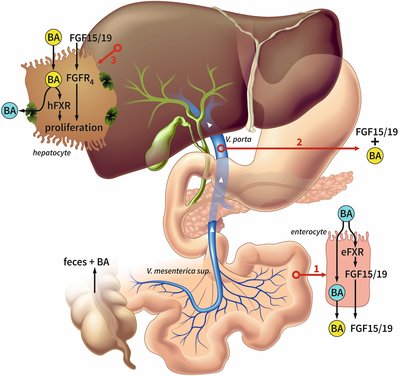
Background: Liver regeneration following partial hepatectomy (PHx) is a complicated process involving multiple organs and several types of signaling networks. The bile acid-activated metabolic pathways occupy an auxiliary yet important chapter in the entire biochemical phenomenon. PHx is characterized by rapid but transient bile acid overload in the liver, which constitutes the first wave of proliferative signaling in the remnant hepatocytes. Bile acids trigger hepatocyte proliferation through activation of several nuclear receptors. The enterocytes reabsorb the bile acids, which results in the activation of farnesoid X receptor (FXR), the consequent excretion of fibroblast growth factor (FGF)19/FGF15, and its release into the enterohepatic circulation. FGF19/FGF15 subsequently binds to its cognate receptor, fibroblast growth factor receptor 4 (FGFR4) complexed with β-klotho, on hepatocytes, which initiates the second wave of proliferative signaling. Because bile acids are potentially toxic, the remnant hepatocytes must resolve the potentially detrimental state of bile acid excess. Therefore, the hepatocytes orchestrate a bile acid detoxification and elimination response as a protective mechanism in concurrence with the proliferative signaling. The response results in the excretion of (biotransformed) bile acids into the canalicular system, causing the bile acids to end up in the intestine.
Relevance for patients: Recently, FXR agonists have been shown to promote regeneration via the gut-liver axis. This type of pharmacological intervention may prove beneficial for patients with hepatobiliary malignancies undergoing PHx. In light of these developments, this review provides an in-depth account of the pathways that underlie post-PHx liver regeneration in the context of bile acid homeostasis in the liver and the gut-liver signaling axis.
DOI: http://dx.doi.org/10.18053/jctres.04.201801.001
Author affiliation
1 Department of Experimental Surgery, Academic Medical Center, University of Amsterdam, Amsterdam, the Netherlands
2 Oncogene Biology Laboratory, Francis Crick Institute, London, United Kingdom
3 Center for Engineering in Medicine, Department of Surgery, Massachusetts General Hospital, Harvard Medical School, Boston, Massachusetts, United States
4 Shriners Hospitals for Children, Boston, Massachusetts, United States
5 Department of Pharmacology, Toxicology and Therapeutics, University of Kansas Medical Center, Kansas City, United States
6 Liver Research Group, Australian National University Medical School at the Canberra Hospital, Canberra, Australian Capital Territory, Australia
7 Membrane Biochemistry and Biophysics, Bijvoet Center for Biomolecular Research, Institute of Biomembranes, Utrecht University, Utrecht, the Netherlands
*Corresponding author:
Tiangang Li
Department of Pharmacology, Toxicology & Therapeutics, Kansas University Medical Center, University of Kansas, Kansas City, KA, United States
Email: tli@kumc.edu
Michal Heger
Department of Experimental Surgery, Academic Medical Center, University of Amsterdam,Meibergdreef 9, 1105 AZ Amsterdam, the Netherlands
Email: m.heger@amc.uva.nl
Handling editor:
Yao Liu
Membrane Biochemistry and Biophysics, Utrecht University, the Netherlands

