-
Home
-
About JCTR
-
Gold Open Access
-
Issues
-
Editorial board
-
Author guidelines
-
Publication fees
-
Online first
-
Special issues
-
News
-
Publication ethics
-
Partners
-
Submit your manuscript
-
Submit your review report
-
Editorial Office
-

This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License. ISSN print: 2382-6533 ISSN online: 2424-810X
Volume 5 Issue 1
Circulating extra-cellular RNAs, myocardial remodeling, and heart failure in patients with acute coronary syndrome
Khanh-Van Tran, Kahraman Tanriverdi, Gerard P. Aurigemma, Darleen Lessard, Mayank Sardana, Matthew Parker, Amir Shaikh, Matthew Gottbrecht, Zachary Milstone, Selim Tanriverdi, Olga Vitseva, John F. Keaney, Catarina I. Kiefe, David D. McManus, Jane E. Freedman
Tran et al., J Clin Transl Res 2019; 5(1): 3
Published on June 8, 2019
Abstract
Background: Given high on-treatment mortality in heart failure (HF), identifying molecular pathways that underlie adverse cardiac remodeling may offer novel biomarkers and therapeutic avenues. Circulating extracellular RNAs (ex-RNAs) regulate important biological processes and are emerging as biomarkers of disease but less is known about their role in the acute setting, particularly in the setting of heart failure.
Methods: We examined the ex-RNA profiles of 296 acute coronary syndrome (ACS) survivors enrolled in the Transitions, Risks, and Actions in Coronary Events Center for Outcomes Research and Education (TRACE-CORE) Cohort. We measured 374 ex-RNAs selected a priori, based on previous findings from a large population study. We employed a two-step, mechanism-driven approach to identify ex-RNAs associated with echocardiographic phenotypes (left ventricular [LV] ejection fraction, LV mass, LV end-diastolic volume, left atrial dimension and left atrial volume index) then tested relations of these ex-RNAs with prevalent HF (N=31, 10.5%). We performed further bioinformatics analysis of microRNA predicted targets’ genes ontology categories and molecular pathways.
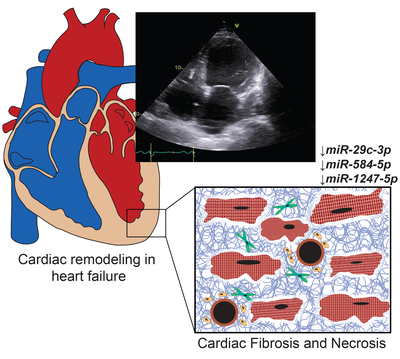
Results: We identified forty-four ex-RNAs associated with at least one echocardiographic phenotype associated with HF. Of these forty-four exRNAs, miR-29-3p, miR-584-5p, and miR-1247-5p were also associated with prevalent HF. The three microRNAs were implicated in the regulation p53 and TGF- signaling pathways and predicted to be involved in cardiac fibrosis and cell death; miRNA predicted targets were enriched in gene ontology categories including several involving the extracellular matrix and cellular differentiation.
Conclusions: Among ACS survivors, we observed that miR-29-3p, miR-584-5p, and miR-1247-5p were associated with both echocardiographic markers of cardiac remodeling and prevalent HF.
Relevance for patients: miR-29c-3p, miR-584-5p, and miR-1247-5p were associated with echocardiographic phenotypes and prevalent HF and are potential biomarkers for adverse cardiac remodeling in heart failure.
DOI: http://dx.doi.org/10.18053/jctres.05.201901.003
Author affiliation
1 Department of Medicine, University of Massachusetts Medical School, Worcester, MA, United States
2 Department of Population and Quantitative Health Sciences, University of Massachusetts Medical School, Worcester, MA, United States
*Corresponding author
Khanh-Van Tran, MD, PhD, Cardiovascular Fellow, Department of Medicine
University of Massachusetts Medical School
368 Plantation Street
Worcester, MA 01655, United States
Email: khanh-van.tran@umassmemorial.org
Handling editor:
Michal Heger
Department of Pharmaceutics, Utrecht University, the Netherlands
Department of Pharmaceutics, Jiaxing University Medical College, Zhejiang, China

