-
Home
-
About JCTR
-
Gold Open Access
-
Issues
-
Editorial board
-
Author guidelines
-
Publication fees
-
Online first
-
Special issues
-
News
-
Publication ethics
-
Partners
-
Submit your manuscript
-
Submit your review report
-
Editorial Office
-

This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License. ISSN print: 2382-6533 ISSN online: 2424-810X
Volume 5 Issue 1
The effect of human amniotic epithelial cells on urethral stricture fibroblasts
Sanjay Gottipamula, Sudarson Sundarrajan, Kumar Chokalingam, K. N Sridhar
Gottipamula et al., J Clin Transl Res 2019; 5(1): 4
Published on July 21, 2019
Abstract
Background: Urethral stricture disease (USD) is effectively managed by buccal mucosa urethroplasty. Lack of adequate healthy buccal mucosa has led to the use of autologous tissue engineered buccal mucosa grafts. Such grafts are costly, not easily scalable and recurrence of the stricture is still a problem. Hence there is a requirement for cost effective, scalable cells with innate anti-fibrotic properties which seem to be fulfilled by human amniotic epithelial cells (HAMECs). The effect of HAMECs on USD is unknown.
Aim: The effect of HAMECs conditioned media (HAMECs-CM) on human urethral stricture fibroblast (USF) cells.
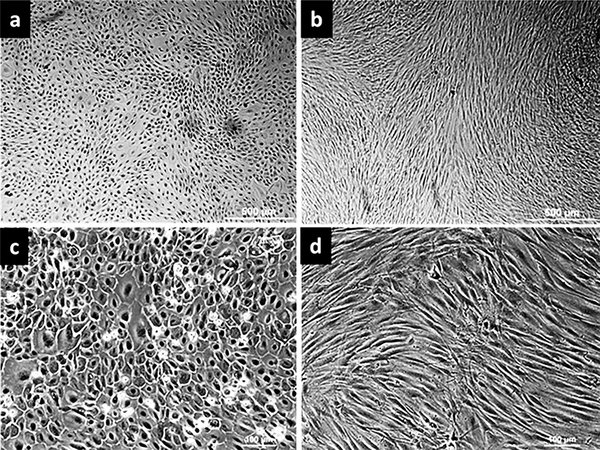
Methods: USF cells were derived from six patients undergoing urethroplasty. HAMECs were derived from one placenta after delivery. The effect of HAMECs-CM on USF cell migration was observed using a standard in vitro scratch assay over a period of 3 days. The effect of HAMECs-CM on the expression levels of markers alpha smooth muscle actin (αSMA) and tissue inhibitor of metalloproteinases (TIMP-1) in USF cells were also examined.
Results: The HAMECs-CM suppressed the migration of USF cells in in vitro scratch assay. The HAMECs-CM consistently downregulated αSMA, but not TIMP-1.
Conclusion: HAMECs have shown anti-fibrotic activity on USF cells in this in vitro study.
Relevance for patients: HAMECs could serve as an alternative cell source for tissue-engineered urethroplasty.
DOI: http://dx.doi.org/10.18053/jctres.05.201901.004
Author affiliation
1. Sri Research for Tissue Engineering Pvt. Ltd, Shankara Research Centre, Rangadore Memorial Hospital, Bangalore, India.
2. Cancyte Technologies Pvt. Ltd, Rangadore Memorial Hospital, Bangalore, India.
*Corresponding author
K. N Sridhar
Sri Research For Tissue Engineering Private Limited, Sri Shankara Research Center, Rangadore Memorial Hospital, 1st Cross, Shankarapuram, Bangalore-560 004, India
Tel: +91-80-41076759, Email: knsridhar@sr-te.com
Handling editor:
Michal Heger
Department of Pharmaceutics, Utrecht University, the Netherlands
Photonanomedicine, Jiaxing University Medical College, Zhejiang, China

